Isovaleric acidaemia is caused by a genetic mutation that disrupts how the body breaks down leucine, an essential amino acid found in protein-rich foods. This inherited metabolic disorder results from a deficiency in an enzyme needed to safely process leucine. Specifically, mutations in the IVD gene affect the enzyme isovaleryl-CoA dehydrogenase, causing toxic substances like isovaleric acid to build up. These toxins lead to the symptoms and complications of the disease.
This section explains the genetic basis, inheritance pattern, biochemical effects, and environmental triggers behind isovaleric acidaemia.
1. Genetic Mutation in the IVD Gene
The root cause lies in mutations of the IVD gene on chromosome 15. This gene provides instructions to make isovaleryl-CoA dehydrogenase, a mitochondrial enzyme crucial for breaking down isovaleryl-CoA, a leucine metabolite.
Mutations may cause:
- Complete loss of enzyme activity
- Severely reduced enzyme function
- Enzyme that is present but works poorly
Over 60 mutations have been found, including missense, nonsense, and frameshift types. The mutation type often influences whether symptoms appear early and severe or later and milder. Even siblings with the same mutation can show different symptom severity, indicating other genetic or environmental influences.
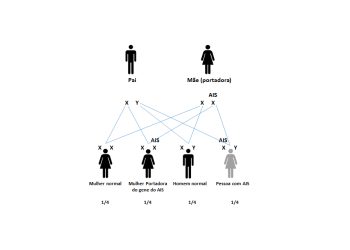
2. Autosomal Recessive Inheritance
Isovaleric acidaemia follows an autosomal recessive pattern. A child must inherit two faulty copies of the IVD gene—one from each parent—to develop the disease. Parents who carry one defective gene usually show no symptoms.
For two carrier parents:
- Each pregnancy has a 25% chance of an affected child
- A 50% chance the child will be a carrier
- A 25% chance the child inherits two normal genes
This inheritance explains why the disorder can appear unexpectedly. Carrier testing and genetic counselling help families understand their risks.
3. Disruption of Leucine Metabolism
Leucine is a branched-chain amino acid in many protein foods like meat, dairy, and nuts. The body breaks it down in steps, one involving isovaleryl-CoA dehydrogenase.
When this enzyme is deficient:
- Isovaleryl-CoA builds up and turns into toxic isovaleric acid
- Toxic compounds like isovalerylglycine and isovalerylcarnitine accumulate in blood and tissues
- These toxins disrupt cell function, especially in the liver, brain, and muscles
- Toxic overload causes metabolic crises with vomiting, lethargy, seizures, and coma, especially during stress or high protein intake
4. Triggers and Environmental Stressors
Though genetic, isovaleric acidaemia symptoms often flare during external stress:
- Infections or fever: Raise metabolic demand and overwhelm detox capacity
- Fasting: Causes the body to break down muscle protein, releasing more leucine
- High-protein diets: Increase leucine load beyond what metabolism can handle
- Surgery or trauma: Physical stress can destabilize metabolism if energy supply is inadequate
These triggers pose higher risk for infants and young children, requiring careful management.
5. Genotype–Phenotype Correlation
Research shows some link between mutation type and disease severity:
- Null mutations often cause the severe neonatal form
- Missense mutations may allow residual enzyme activity, leading to milder, intermittent symptoms
Still, symptom severity can vary even with the same mutation, influenced by other genes, metabolism, and environment. This variability highlights the need for personalized care.
6. Carrier Frequency and Ethnic Variability
Isovaleric acidaemia is rare but carrier rates differ by population. Some communities show higher carrier frequencies due to founder effects, such as:
- Amish and Mennonite groups in North America
- Certain regions in Saudi Arabia and India
- Some Central European populations
Knowing these patterns helps tailor screening and education efforts.
7. Prenatal and Preimplantation Genetic Diagnosis
Couples with known mutations can use genetic counselling for reproductive planning. Options include:
- Prenatal testing via chorionic villus sampling (CVS) or amniocentesis
- Preimplantation genetic diagnosis (PGD) with IVF to select embryos without the mutation
These methods enable early detection and informed decision-making.
Conclusion
Isovaleric acidaemia results from mutations in the IVD gene causing enzyme deficiency and toxic buildup during leucine breakdown. It follows an autosomal recessive pattern, with symptoms often triggered by metabolic stress, especially in infants. Though rare, the condition can be managed well through early diagnosis, genetic counselling, dietary control, and medical care. Understanding its genetic and biochemical roots paves the way for better outcomes and support for affected families.